What is a hydrocarbon? Definition and examples
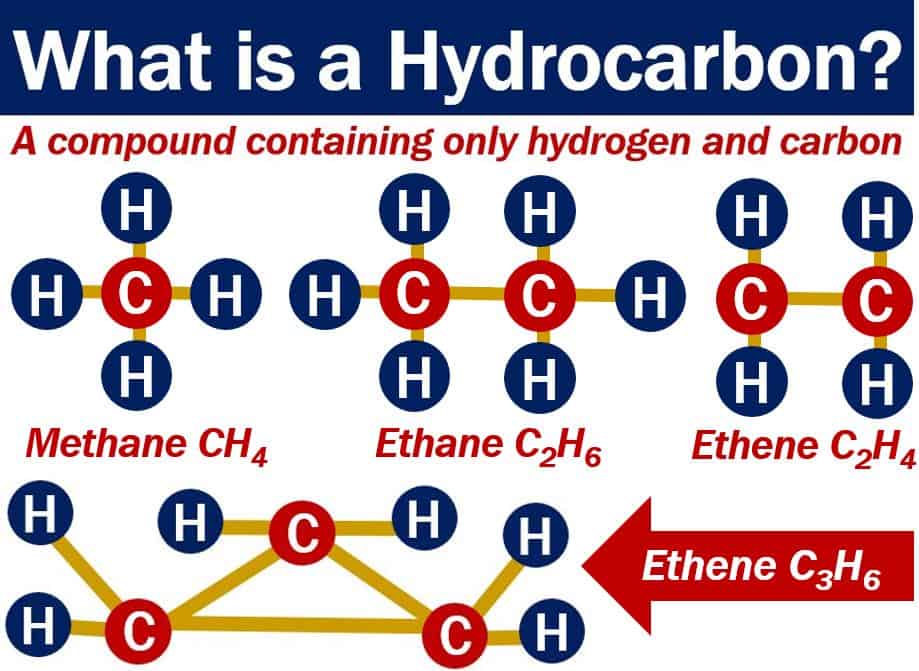
Hydrocarbons are compounds made entirely of carbon and hydrogen atoms.
Benzene, paraffin, ethane, propane, hexane, naphthalene, and methane, for example, are hydrocarbons. We can find hydrocarbons in natural gas, crude oil, coal, and plant life.
The simplest hydrocarbon is methane. Methane consists of one carbon atom with four hydrogen atoms stuck to it.
Our solvents and fuels
We use hydrocarbons as solvents and fuels. We also use them as raw materials for pesticides, plastics, dyes, rubbers, explosives, and hundreds of different products.
In fact, petroleum, from which we get diesel and gasoline (UK: petrol), is a mixture of many different hydrocarbons.
According to Britannica.com, the term refers to:
“Any of a class of organic chemical compounds composed only of the elements carbon (C) and hydrogen (H). Hydrocarbons are the principal constituents of petroleum and natural gas.”
“They serve as fuels and lubricants as well as raw materials for the production of plastics, fibers, rubbers, solvents, explosives, and industrial chemicals.”
Without hydrocarbons, our current oil economy would not exist. In fact, hydrocarbons are crucial in many biochemical processes and are components of many organic molecules.
When the air contains less than 0.1 parts per million of hydrocarbons we call it zero air.
Hydrocarbons in nature
Most hydrocarbons we find naturally occur in crude oil. There are three main sources of natural hydrocarbons: petroleum, coal, and natural gas.
Apart from being present in fossil fuels, they also exist naturally in plants. Green leaves and carrots, for example, contain carotene. Carotene, a red or orange plant pigment, is a terpenoid hydrocarbon.
In outer space, the largest hydrocarbon molecules are amino acids. On Earth, however, those amino acids hooked up with each other to create protein molecules.
A protein molecule, in fact, may include hundreds of amino acids in many different combinations.
Hydrocarbons come in many forms
Hydrocarbons can be:
- Gases, such as propane or methane.
- Liquids, including benzene or hexane.
- Waxes or Low-Melting Solids, such as naphthalene and paraffin wax.
- Polymers, including polystyrene, polypropylene, and polyethylene. A polymer is a macromolecule, i.e., a large molecule, comprising several repeated sub-units.
In living cells
Protein molecules are present in every living cell on Earth. Therefore, all living cells are made out of hydrocarbons. hydrocarbons are integral to the lipid structures that form cell membranes, playing a crucial role in maintaining cell integrity and facilitating biological functions.
Our bodies, fishes, trees, seaweed, cheese, and milk all contain organic molecules built upon carbon frameworks, many of which are hydrocarbons.
In fact, anything that lives or once lived is made from hydrocarbons. Therefore our food, as well as rubber, alcohol, and even antibiotics contain hydrocarbons.
Hydrocarbon families
We group hydrocarbon into homologous series or families. There are five main homologous groups. In each family, the hydrocarbons have a general formula and similar chemical properties. They behave in similar ways. The five families are:
Alkanes
Of all the hydrocarbons, alkanes are the simplest. Examples include:
- Methane CH4
- Ethane C2H6
- Propane C3H8
- Butane C4H10
- Pentane C5H12
- Hexane C6H14
- Heptane C7H16
- Octane C8H18
- Nonane C9H20
- Decane C10H22
Alkenes
Examples of alkenes include:
- Ethene C2H4
- Propene C3H6
- Butene C4H8
- Pentene C5H10
- Hexene C6H12
- Heptene C7H14
- Octene C8H16
- Nonene C9H18
- Decene C10H20
Alkynes
Examples of alkynes include:
- Ethyne (Acetylene) C2H2
- Propyne C3H4
- Butyne C4H6
- Pentyne C5H8
- Hexyne C6H10
- Heptyne C7H12
- Octyne C8H14
- Nonyne C9H16
- Decyne C10H18
Cycloalkanes
Examples of Cycloalkanes include:
- Cyclopropane C3H6
- Cyclobutane C4H8
- Cyclopentane C5H10
- Cyclohexane C6H12
- Cycloheptane C7H12
- Cycloctane C8H16
- Cyclononane C9H18
- Cyclodecane C10H20
Alkadiene
Alkadienes are isomers of Alkynes. Alkadienes have two carbon to carbon double bonds.
An isomer is a molecule with the same molecular formula of another molecule. However, the isomer has a different chemical structure. In other words, isomers contain the same number of each element’s atoms, but they are arranged differently.
Hydrocarbon vocabulary
There are many compound phrases in the English language that contain the word “hydrocarbon.” Let’s have a look at some of them:
-
Hydrocarbon Fuels
Energy sources composed of hydrocarbons.
Example: “Hydrocarbon fuels, such as gasoline, power most of our vehicles.”
-
Hydrocarbon Chains
Molecular structures consisting of a sequence of hydrocarbons.
Example: “Plastics are made of long hydrocarbon chains that give them flexibility.”
-
Hydrocarbon Emissions
Gases released from burning hydrocarbon compounds.
Example: “Regulations aim to reduce hydrocarbon emissions from factories.”
-
Hydrocarbon Reserves
Natural deposits of hydrocarbons in the earth.
Example: “The country’s economy relies heavily on its hydrocarbon reserves.”
-
Hydrocarbon Extraction
The process of obtaining hydrocarbons from natural resources.
Example: “Advancements in hydrocarbon extraction techniques have increased oil production.”
-
Aromatic Hydrocarbons
Hydrocarbons known for their stable ring structures.
Example: “Benzene, a common solvent, is one of the simplest aromatic hydrocarbons.”
Video – What are Hydrocarbons?
This educational video, from our sister channel on YouTube – Marketing Business Network, explains what ‘Hydrocarbons’ are using simple and easy-to-understand language and examples.

