What is carbon capture and storage (CCS)?
Carbon capture and storage (CCS) refers to the process of capturing the greenhouse gas carbon dioxide (CO2) and transporting it to a safe location to be stored in.
The International Energy Agency (IEA) says that carbon capture is an important emissions reduction technology that can be applied across the energy system. The IEA said in a report that CCS could contribute to a 19% reduction in global CO2 emissions by 2050 and that it could cost an estimated 70% more to fight climate change without CCS.
According to the Carbon Capture & Storage Association, there are three key parts of CSS:
- Capturing carbon dioxide released during the burning of fossil fuels or as a result of industrial processes.
- Transporting the captured carbon dioxide by pipeline or ship for storage at a safe location.
- Securely storing the captured carbon dioxide. It is typically stored in porous geological formations several km below the earth’s surface.
Capturing carbon dioxide
Carbon capture is the separation of carbon dioxide from gases. Absorption, adsorption, or membrane gas separation technologies are typically used to separate carbon dioxide out of air.
There are three different ways of capturing carbon dioxide: post-combustion, pre-combustion, and oxyfuel combustion.
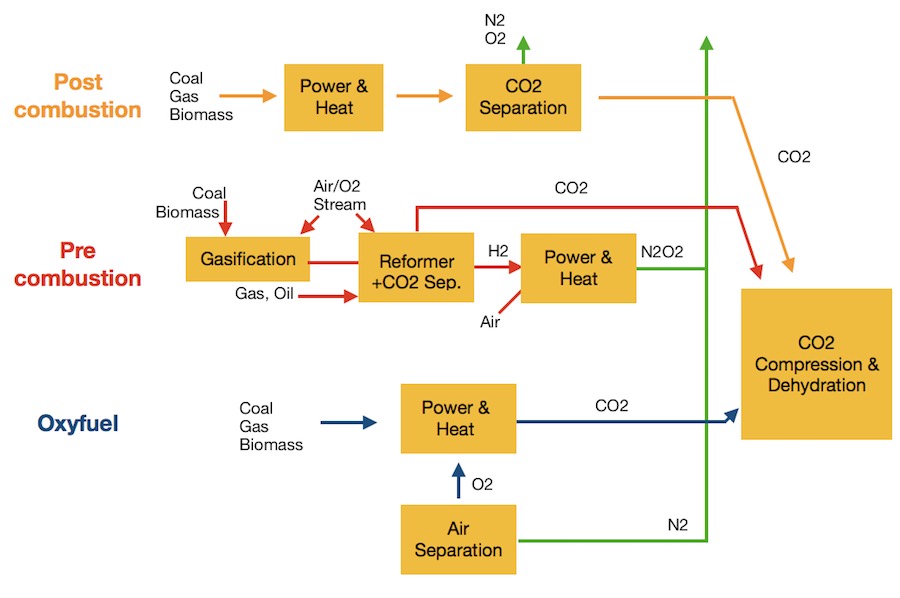
Pre-combustion capture
This process involves removing carbon dioxide from fuel sources before combustion is completed. Gasification, partial oxidation or steam reforming technology can be used to convert the fuel source to a syngas (mostly a mixture of carbon monoxide and hydrogen).
The carbon monoxide can then be converted into carbon dioxide through a shift conversion process which also produces a stream of hydrogen, which can be used as a fuel.
Post-combustion capture
Carbon dioxide is removed after combustion of the fossil fuel. The carbon dioxide is typically absorbed it in a suitable solvent and then liberated and compressed for transportation and storage.
Oxyfuel combustion
This is the process of burning a hydrocarbon fuel source with almost pure oxygen instead of air.
The gaseous combustion reaction product is almost pure carbon dioxide which can be purified for sequestration through physical separation processes.
Transporting the captured carbon dioxide
 Once the carbon dioxide has been captured it is compressed into liquid state and is then transported by pipeline or ship to a suitable location for storage.
Once the carbon dioxide has been captured it is compressed into liquid state and is then transported by pipeline or ship to a suitable location for storage.
According to the Carbon Capture & Storage Association, “technologies involved in pipeline transportation are the same as those used extensively for transporting natural gas, oil and many other fluids around the world.”
Storing the captured carbon dioxide
According to the LSE Grantham Research Institute on Climate Change and the Environment, once the carbon dioxide is captured it can be pumped several kilometres underground “where it is stored into depleted oil and gas reservoirs, coalbeds or deep saline aquifers, where the geology is suitable.”
Carbon dioxide is injected under pressure at the storage site into a geological formation. The carbon dioxide moves up through the storage site until it reaches an impermeable layer of rock, known as the cap rock. The cap rock keeps the carbon dioxide trapped in the storage formation. This storage mechanism is called “structural storage”.

