What is CBD or Cannabidiol? Definition and examples
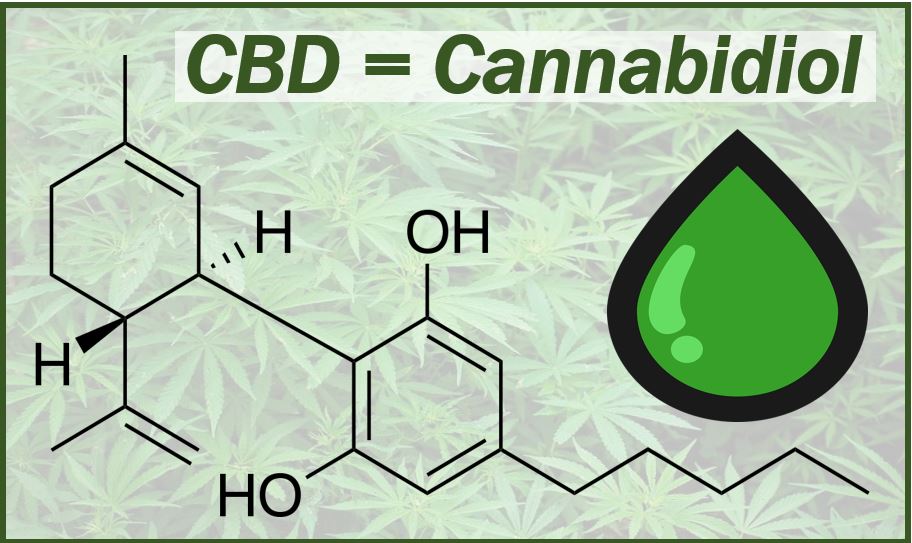
CBD stands for Cannabidiol. Recently, it has become a popular term in the media; especially CBD oil. CBD is one of the active ingredients of marijuana or cannabis. In fact, it is the plant’s second most prevalent active ingredient.
CBD, a phytocannabinoid, was discovered in 1940. Cannabis plants contain 113 cannabinoids that we know of. Cannabidiol is one of those cannabinoids and makes up approximately forty percent of the cannabis plant’s extract.
The extraction of CBD from cannabis plants is a critical process, typically involving methods such as CO2 extraction or solvent extraction, which are designed to isolate the compound while preserving its purity and potency.”
CBD and some other remedies have become increasingly popular especially in North America, Europe, Japan, and Australia.
CBD not a psychoactive drug
Psychoactive substances, when consumed, affect mental processes. We all associate cannabis with getting high (doped up, euphoric). However, CBD does not exhibit any of the high effects on humans. In other words, it is not a psychoactive substance.
According to the World Health Organization:
“In experimental models of abuse liability, CBD appears to have little effect on conditioned place preference or intracranial self-stimulation.”
“In an animal drug discrimination model CBD failed to substitute for THC (tetrahydrocannabinol). In humans, CBD exhibits no effects indicative of any abuse or dependence potential.”
THC is cannabis’ principal psychoactive constituent – it is the principal substance that gives marijuana users a high.
Is CBD a legal drug?
-
United States
Cannabidiol is available in most parts of the USA and many other countries today. Fifty US states have legislation legalizing CBD. However, they do not all have the same degree of restrictions.
In 2015, the Food and Drug Administration (FDA) eased the regulations regarding CBD research and trials.
The FDA is the US regulatory agency in charge of medical devices, drugs (medications), tobacco products, cosmetics, and food. Pharmaceutical companies must get FDA approval before they can market a new drug.
-
United Kingdom
In the UK, CBD is legal as long as it has been derived from an EU-approved industrial hemp strain. These strains contain either no THC or very little. CBD is legal in the UK as long as its THC content does not exceed 0.2%.
Cannabis oil, for example, which has a higher THC content, is illegal in Britain.
-
Canada
In Canada, CBD became legal for therapeutic use. In 2018, it also became legal for recreational use.
- Australia
In Australia, cannabidiol is legal for therapeutic use. It must not contain more than 2% of other cannabinoids such as THC. In 2017, CBD became a schedule 4 drug (from schedule 9), thus making it legally available in the country.
Therapeutic uses
Humans can take CBD by inhalation, as an aerosol spray applied to the inside of the cheek, and orally (by mouth).
-
Epilepsy
Research on CBD treatment for epilepsy has been promising when it is used in combination with conventional drugs. There are some minor side effects.
Some studies also pointed to potential uses of CBD for other neurological disorders. However, further research is required.
In 2018, the US FDA approved Epidiolex for the treatment of Dravet syndrome and Lennox-Gastaut syndrome, two rare types of childhood epilepsy. Epidiolex, which comes as a liquid solution that the patient takes by mouth, contains CBD
Studies showed that Epidiolex reduced the number of seizures in patients with Lennox-Gastaut syndrome and Dravet syndrome by 20-25% and 26%, respectively. The trials were carried out by the makers of Epidiolex, GW Pharmaceuticals.
A study by researchers from the University of California, Davis, used a synthetic form of CBD. The substance showed promising results in the treatment of seizures in laboratory rats. The researchers said that the synthetic cannabinoid alternative is significantly easier to purify than the plant extract. Their study was published in the prestigious journal Nature.
-
Insomnia
Cannabidiol is used to treat anxiety and insomnia. According to some studies, CBD may help patients either fall asleep, remain asleep, or both.
-
Pain
A double-blind, randomized, placebo-controlled clinical trial in 2014 showed that CBD helped patients with peripheral neuropathic pain. People with peripheral neuropathic pain experience weakness, numbness, and pain in their hands, feet, or both.
The study researchers, from the UK and the Czech Republic, wrote about the trial in the European Journal of Pain.
Beyond human medicine, veterinarians are also exploring the benefits of CBD for addressing similar conditions in animals, particularly for pain management and anxiety relief in pets.
Educational Video – What is CBD?
This interesting video comes from our sister channel in YouTube, Marketing Business Network. It explains what “CBD” is using easy to understand language and examples:

