Carbon Dioxide or CO2 is an odorless and colorless gas that is vital for life as we know it. It is a naturally occurring compound composed of one carbon atom covalently double-bonded (sharing electron pairs) to two oxygen atoms. It is also a greenhouse gas.
CO2 exists in our atmosphere as a trace gas at a concentration of approximately 420 ppm (parts per million) or 0.042%. Natural sources include geysers, hot springs and volcanoes – it is freed from carbonate rocks by dissolution in acids and water.
Carbon dioxide is soluble in water – it dissolves in water – and occurs naturally in lakes, rivers, seawater, groundwater, ice caps and glaciers. It is also present in deposits of natural gas and petroleum.
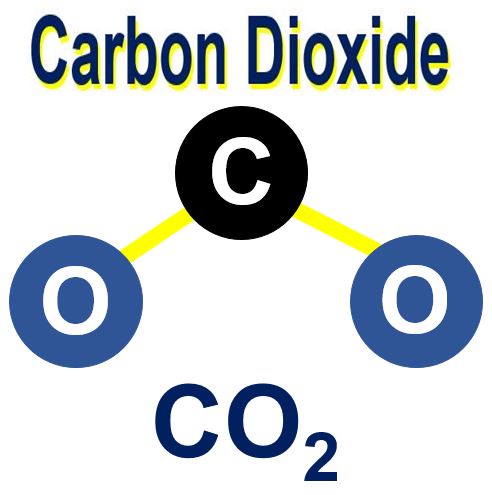
A carbon dioxide molecule consists of one carbon atom and two oxygen atoms.
Carbon dioxide on Earth and in space
CO2 is one of the commonest and simplest molecules in the Universe. There are carbon dioxide molecules in outer space, where they formed in nebulae after supernova explosions.
In space, CO₂ often exists as ice; at standard atmospheric pressure on Earth, solid carbon dioxide (dry ice) sublimates at -78.5°C (-109.3°F). Its triple point (where solid, liquid, and gas can coexist) is -56.4°C (-69.5°F) under higher pressure.
. That is why there is carbon dioxide not just on Earth, but also on Mars, Venus and other planets.
Earth’s surface is considerably warmer than space, so our carbon dioxide melted and became a gas – the CO2 molecules float in the air. Even though a tiny percentage of our atmosphere consists of carbon dioxide, if it did not exist there would be no life on Earth.
Additionally, carbon dioxide plays a crucial role in regulating Earth’s temperature by absorbing infrared radiation, which helps maintain the planet’s energy balance and contributes to the greenhouse effect.
Plants make their cells principally out of carbon. They get their carbon by breathing in CO2 and breaking off the oxygen, which they breathe out (and keep the carbon). So, plants are made of the carbon in carbon dioxide molecules, while the oxygen becomes the oxygen we and other animals breathe.
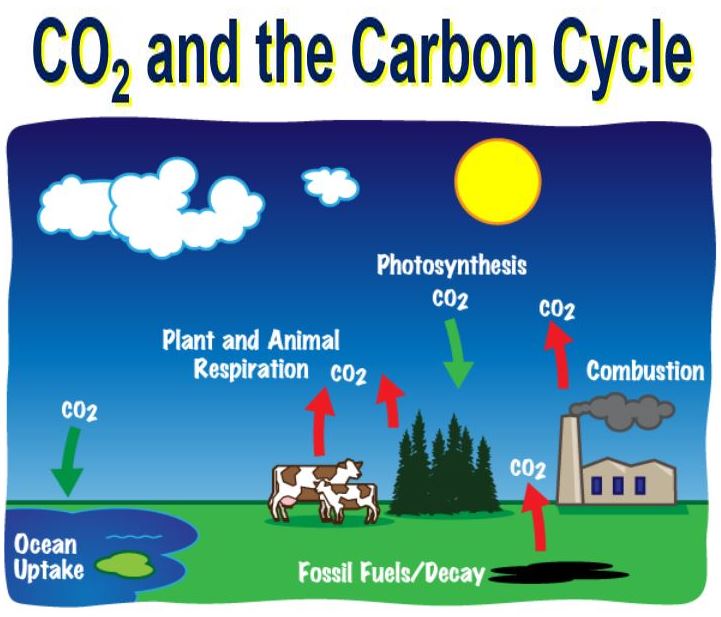
CO2 is a major part of the carbon cycle.
The carbon returns to the atmosphere when a plant dies and decays, or burns – it mixes with oxygen to form carbon dioxide molecules again.
Greenhouse gases and the greenhouse effect
Greenhouse gases absorb infrared radiation emitted by the Earth’s surface and then re-emit it in all directions, warming the lower atmosphere.
Carbon dioxide, along with methane, water vapour, nitrous oxide and ozone are greenhouse gases – they are responsible for the greenhouse effect.
The greenhouse effect is a natural process in which a planet’s – such as Earth’s – atmosphere traps some of its parent star’s energy, warming it enough to support life. Our parent star is the Sun.
It is thanks to the greenhouse effect that we, other animals, plants and all life forms on Earth exist.
Without methane, CO2 and other greenhouse gases, Earth’s surface temperature would average -18°C (0°F), rather than the current 15°C (59°F) – our days would reach 123°C (253°F), while at night the temperature would plunge to -153°C (-243°F), like on the surface of the Moon.
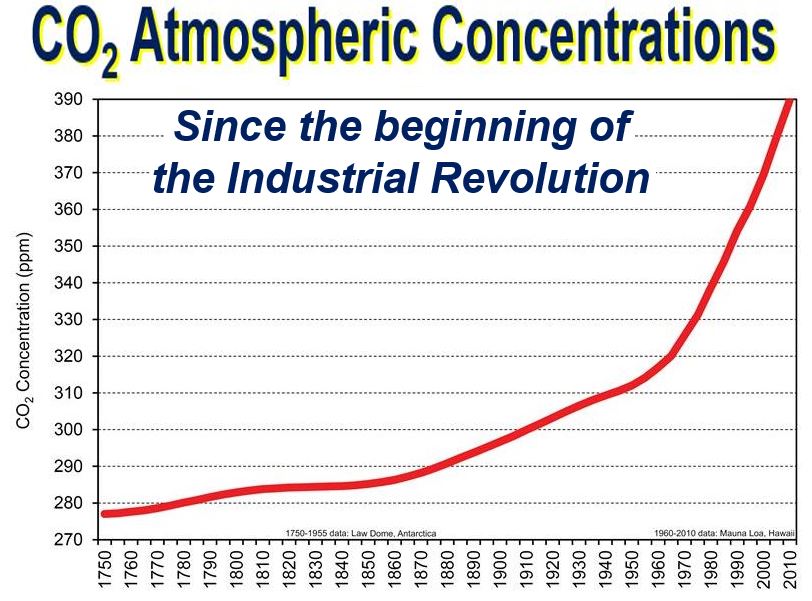
Since the middle of the last century, CO2 concentrations have been increasing at a much faster rate. The graph ends in 2010 – the figure for today is much higher. (Image: gsu.edu)
Carbon dioxide concentrations rising
CO2 concentrations have been ideal for life to thrive on Earth for hundreds of millions of years. Concentrations of the other greenhouse gases have also been ideal. Sometimes they have fluctuated for various reasons, especially before and during some of the mass extinctions.
At the moment, scientists and policymakers across the world are becoming increasingly concerned about CO2 concentrations, which have been rising since 1750 – the beginning of the Industrial Revolution. Carbon dioxide atmospheric concentrations have increased by over 40% since 1750, and will probably continue going up. Global efforts like the Paris Agreement aim to limit global temperature rise by reducing greenhouse gas emissions, including CO₂.
Levels of other greenhouse gases have also risen. Earth’s temperature has increased since the industrial revolution. Experts predict that our planet’s surface and atmosphere will get warmer and warmer. There is a risk that the polar ice caps will melt, which would cause sea levels to rise.
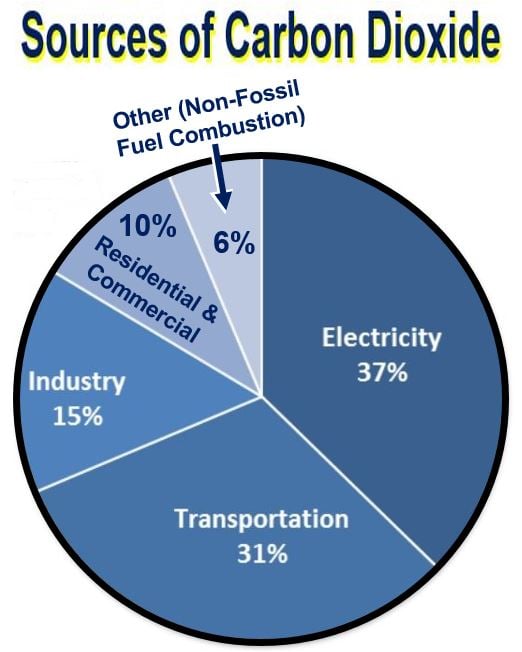
These estimates are just for the United States for the period 1990-2014. (Image: epa.gov)
Millions could end up homeless
Millions of people across the world could end up with nowhere to live if their homes become submerged. Especially those who live in cities and towns on the coast.
CO2 is a heat-trapping greenhouse gas. The more of it there is in Earth’s atmosphere, the warmer it gets. Whenever we burn fossil fuels such as natural gas, oil and coal, we are producing CO2. Apart from fossil fuel use, deforestation and land-use changes also release large amounts of carbon dioxide by reducing the number of trees and plants that absorb CO₂.
Carbon is not only found in the atmosphere. Our oceans store huge quantities of carbon, and so do deposits of coal, oil, natural gas deep underground, and plants. Carbon moves naturally from one part of our planet to another through the carbon cycle.
At the moment, we are adding carbon to the atmosphere faster than the natural processes can remove it. We are adding carbon in the form of in the form of CO2. That is why carbon dioxide concentrations in our atmosphere are increasing, which is causing global warming (climate change).
In recent years, technological advancements have led to the development of carbon capture and storage (CCS) methods, which aim to reduce CO2 emissions by capturing carbon dioxide from industrial processes and storing it underground, thereby mitigating its impact on climate change.
Global warming and climate change do not have the same meaning. Climate change covers a wide range of shifts in climate and weather patterns, including changes in rainfall, more frequent extreme weather events, and ocean acidification. Global warming specifically refers to the overall rise in average surface temperature.
If global temperatures declined, that would be a climate change matter, but not a global warming one. Global warming only involves temperature rises.
Video explanation
This interesting video presentation explains what ‘Carbon Dioxide’ is using simple and easy-to-understand language and examples.
