What are fluorocarbons? Definition and examples
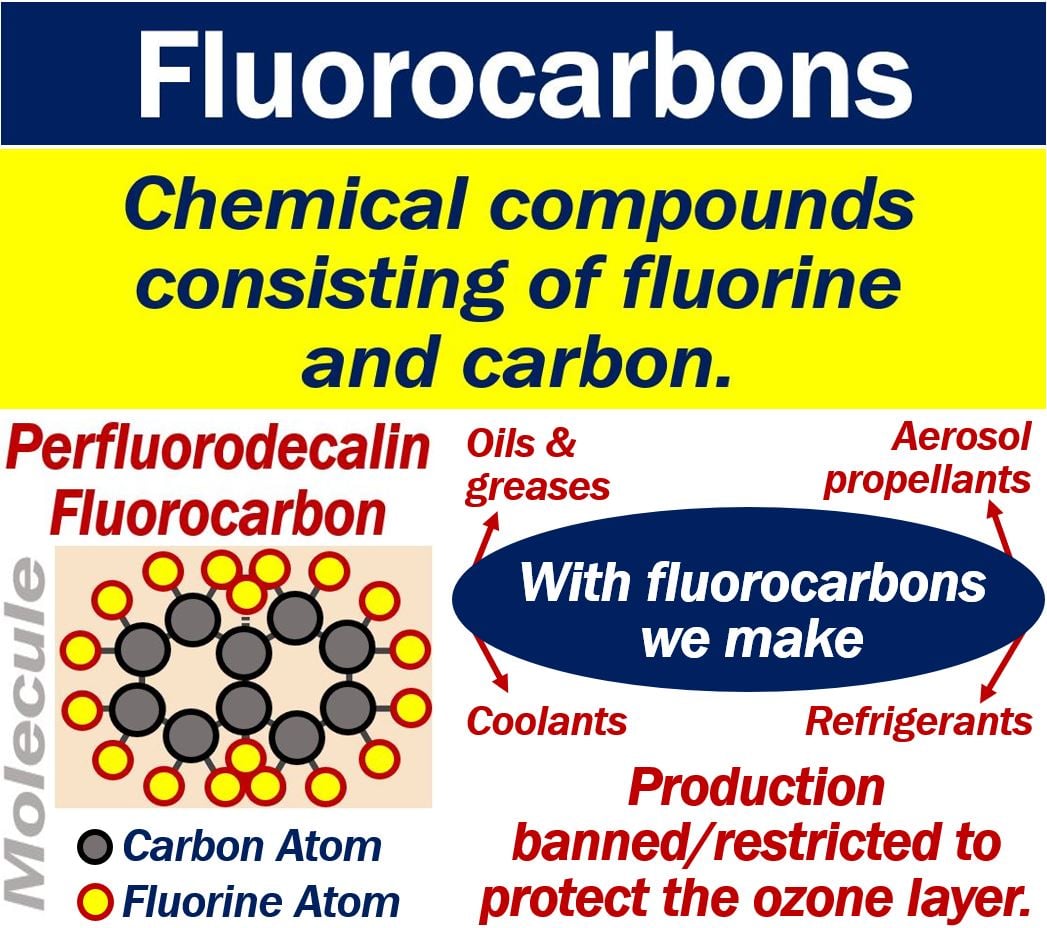
Fluorocarbons of FCs are compounds that contain fluorine and carbon. People also call them perfluorocarbons or PFCs. We use fluorocarbons in the manufacture of coolants, refrigerants, oils, and greases. We also use them in the manufacture of aerosol propellants.
The majority of fluorocarbons are non-reactive and non-flammable. Non-flammable means they do not catch fire easily. Non-reactive means they are not reactive, i.e., don’t react, unless under severe conditions or with a catalyst. FCs are stable at high temperatures and have low toxicity.
Scientists, however, suspect that fluorocarbons cause damage to the ozone layer. That is why their production is either illegal or restricted.
Merriam-Webster defines fluorocarbons as:
“Any of various chemically inert compounds containing carbon and fluorine used chiefly as lubricants, refrigerants, nonstick coatings, and formerly aerosol propellants and in making resins and plastics.”
Fluorocarbons and ozone
Ozone, a gas consisting of three oxygen atoms, is our planet’s natural sunscreen. It absorbs most of the UV radiation that comes from the Sun. UV stands for ultraviolet. This ozone ‘filter’ protects life from radiation that damages DNA.
Fluorocarbons are ozone destroyers. The more ozone we produce, the less ozone there will be protecting us from the Sun’s rays.
Fluorocarbons do not directly affect the ozone layer. FCs are greenhouse gases that help increase the amount of water vapor that gets into the ozone layer. It is the water vapor in the ozone layer that depletes ozone.
Severe ozone depletion
Severe ozone depletion would cause the following problems:
- An increase in the incidence of malignant melanoma (skin cancer) and cataracts among humans.
- Plants would not be able to grow and absorb nutrients properly.
- The amount of phytoplankton in the sea would diminish significantly. Phytoplankton is the foundation of the aquatic food web.
- More UVB radiation would affect the development of amphibians, shrimp, and several fish species. It would also affect the development of hundreds of other species of marine animals.
- An increase in UVB radiation would alter the sources and sinks of greenhouse gases. Put simply; levels of gases that contribute to global warming would increase.
Global warming refers to the consistent increase in the average temperatures of our oceans and atmosphere.
– Many of the polymers we make, i.e., synthetic polymers, would break down earlier outdoors. In other words, they would not last as long.
Fluorocarbons – anesthetics
During the 1960s, there was a lot of interest in using fluorocarbons in hospital procedures. Specifically, using FCs as anesthetics.
Ethylene and cyclopropane, researchers found, elicited surgical anesthesia when humans inhaled them in sufficient concentrations.
However, after some research and limited trials, scientists abandoned the idea. We use some fluorocarbon derivatives in anesthetics today.

