Ozone – definition of meaning
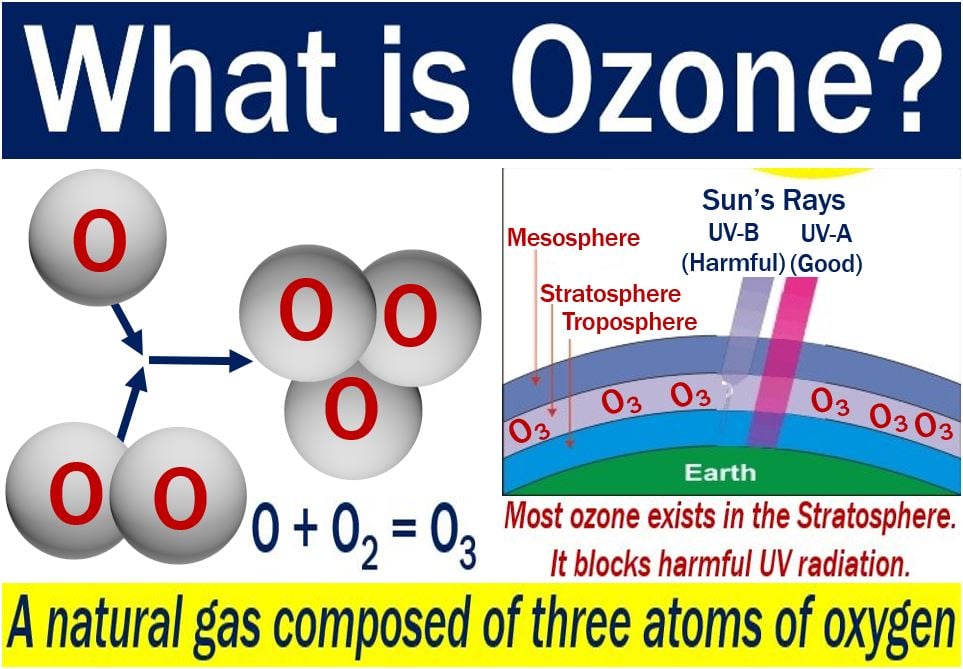
Ozone is a gas consisting of three oxygen atoms. Its chemical formula is O3. The gas occurs in trace amounts in the stratosphere. The stratosphere is the second major atmospheric layer, just above the troposphere. The stratosphere extends in altitude from approximately 8 to 30 miles (12.8 to 48.8 kilometers) high.
O3 protects us from the Sun’s UV radiation. UV stands for ultraviolet. In fact, O3 protects all life forms. In the troposphere, chemical reactions from pollutants create O3. Examples of the pollutants include gasoline vapors, vehicle exhaust, and other emissions.
High levels of ozone at ground level are toxic to humans, other animals, and plants.
According to the NASA Ozone Watch at the Goddard Space Flight Center:
“Ozone is a gas made up of three oxygen atoms (O3). It occurs naturally in small (trace) amounts in the upper atmosphere (the stratosphere).”
“Ozone protects life on Earth from the Sun’s ultraviolet (UV) radiation.”
‘Good’ ozone
Most of the O3 in our atmosphere sits in the stratosphere; in fact, 90% of it is there. Its natural level in the stratosphere is a result of a balance between O3, which the Sun creates, and chemical reactions which destroy it.
When sunlight splits O2 apart into single oxygen atoms, the result is O2 or O3.
The single atoms can re-join to create O2, which is the oxygen we breathe. The single atoms can also join with O2 to create O3.
Bromine, chlorine, hydrogen, and nitrogen destroy O3 when they react with it. Some of the O3-destroying molecules occur naturally. However, there are others that human activity has created.
How much ozone is there?
There are about three million metric tons of ozone in our atmosphere. Three metric tons sounds like a lot, but it only represents 0.00006% of the atmosphere.
Peak O3 concentrations occur at an altitude of approximately 20 miles (32 kilometers) above the Earth’s surface. At that height, the O3 concentration may reach 0.0015% or fifteen parts per million.
O3 absorbs UV radiation
The ozone in our atmosphere absorbs most of the Sun’s UV radiation. In fact, the Earth’s surface would be sterile, i.e., lifeless, without O3.
O3 filters out most of the UV-b radiation and all of the most energetic UV-c radiation. It only screens approximately half of the UV-a radiation.
High exposure to UV-b and UV-a radiation causes sunburn and raises the risk of eye damage and skin cancer.
Chlorofluorocarbons (CFCs), which humans produce, have led to higher rates of O3 destruction in our atmosphere. Chlorofluorocarbon molecules contain carbon, fluorine, and chlorine.
Fluorocarbon molecules contain carbon and fluorine. Fluorocarbons also deplete ozone.
In fact, human activity has upset the natural balance of ozone. In other words, we are causing a reduction of stratospheric ozone levels.
As O3 levels decline, the amount of harmful ultraviolet radiation hitting the Earth’s surface has increased.
When researchers mention the ‘ozone hole,’ they are talking about ‘good’ O3 destruction.
Scientists warn that a massive solar flare could seriously deplete out atmospheric ozone.
Bad ozone
When O3 levels at ground level, i.e., in the troposphere, rise, human health suffers. In fact, all life on Earth suffers.
Human exposure to ozone levels higher than 70 parts per billion for over eight hours is bad for the health. Many large cities across the world frequently exceed this level.
Harmful O3 levels can aggravate asthma and emphysema, and cause lung and throat irritation.
O3 at ground level forms when nitrogen oxide reacts with volatile organic compounds. Vehicles and industry produce nitrogen oxide gases.
Paint thinners and other substances we produce, evaporate easily into the air as volatile organic compounds.
Video – Good and bad ozone
This University of Illinois Extension video explains the difference between ‘good’ and ‘bad’ O3. During the spring and summer, news reports tell us that high levels of the gas harm us.
However, in the fall, the media warns that the loss of O3 in the upper atmosphere causes health problems. How can they both affect our health?

