Kryptonite synthesis – making kryptonite – is now possible, say Polish scientists, who have found how to manufacture the first binary compound of oxygen and krypton – a krypton oxide. So, if an evil version of Superman came to do us harm, we would be able today to weaken him.
Well, perhaps not – a krypton oxide may not be as effective in weakening Superman, his family and other beings born on the planet Krypton, as kryptonite itself.
The chemists, from the Institute of Physical Chemistry of the Polish Academy of Sciences (IPC PAS), say this exotic substance can be synthesized under extremely high pressure, and producing it successfully is well within the capabilities of most modern laboratories.
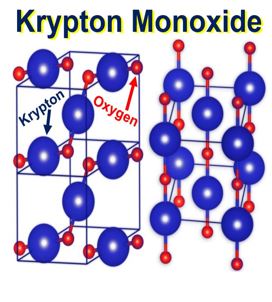
 The crystal structures of krypton monoxide KrO: more stable on the left, less stable on the right. The krypton atoms are coloured blue, and the oxygen atoms are red. (Image: Institute of Physical Chemistry)
The crystal structures of krypton monoxide KrO: more stable on the left, less stable on the right. The krypton atoms are coloured blue, and the oxygen atoms are red. (Image: Institute of Physical Chemistry)
Kryptonite formation likely needed extremely high pressure
Crystals of kryptonite, a substance Superman and his race feared for their lives, were said to have been created within the planet Krypton, probably under extremely high pressure.
Real krypton, the progenitor of the name, is an element with the atomic number 36. It is a noble gas said to be incapable of forming stable chemical compounds. The noble gases are in Group 0, on the far right of the periodic table, and include helium, neon, argon, krypton, xenon, and radon – they are all chemically unreactive gases.
However, two theoretical chemists wrote in the academic journal Scientific Reports (citation below) that it is possible to synthesize a new crystalline material in which krypton atoms would be chemically bonded with another element.
A compound of krypton with oxygen
Co-author, Dr. Patrick Zaleski-Ejgierd (IPC PAS), said:
“The substance we are predicting is a compound of krypton with not nitrogen, but oxygen. In the convention of the comic book it should, therefore, be called not so much kryptonite as kryptoxide. So if Superman’s reading this, he can stay calm – at the moment there’s no cause for panic!”
 Dr. Patrick Zaleski-Ejgierd (left) and Pawel M. Lata testing the krypton monoxide they synthesized on Superman. Actually, not the man himself, they could not find him. Does dressing up like him count as a scientific test? (Image: Institute of Physical Chemistry. Credit: Grzegorz Krzyzewski)
Dr. Patrick Zaleski-Ejgierd (left) and Pawel M. Lata testing the krypton monoxide they synthesized on Superman. Actually, not the man himself, they could not find him. Does dressing up like him count as a scientific test? (Image: Institute of Physical Chemistry. Credit: Grzegorz Krzyzewski)
“Our krypton monoxide, KrO, probably does not exist in nature. According to current knowledge, deep in the interiors of planets, that is, the only place where there is sufficient pressure for its synthesis, oxygen does not exist, nor even more so, does krypton.”
Krypton compounds have been produced before, in the lab under cryogenic conditions. However, they were only single, linear and small molecules of the hydrogen-carbon-krypton-carbon-hydrogen type.
Dr. Zaleski-Ejgierd and PhD student Pawel M. Lata, both from the Institute of Physical Chemistry, wondered whether conditions might exist where krypton would not only bond chemically with another element, but also form an extensive and stable crystal lattice.
Scientists used Density Functional Theory
Their quest, which was funded by an OPUS grant from the Polish National Science Centre, involved the two scientists using genetic algorithms and models built on the Density Functional Theory (DFT).
DFT is a computational quantum mechanical modelling method used in chemistry, physics and materials science to investigate electronic structure of many-body systems, especially atoms, molecules, and the condensed phases.
In the field of solid state physics, DFT has for several years been a basic tool for the description and study of the world of chemical molecules.
 Kryptonite is a substance from the Superman comic books and subsequent TV series and movies. The material, usually shown as having been formed from the remains of his native planet of Krypton, weakens Superman, and is even capable of killing him.
Kryptonite is a substance from the Superman comic books and subsequent TV series and movies. The material, usually shown as having been formed from the remains of his native planet of Krypton, weakens Superman, and is even capable of killing him.
Lata said:
“Our computer simulations suggest that crystals of krypton monoxide will be formed at a pressure in the range of 3 to 5 million atmospheres. This is a huge pressure, but it can be achieved even in today’s laboratories, by skillfully squeezing samples in diamond anvils.”
Crystal lattices are built from molecules or atoms arranged in space in an orderly manner. The smallest repetitive fragment of these structures – the basic ‘building block’ – is called the unit cell.
Table salt crystal unit cells have the shape of a cube – the chlorine and sodium atoms – arranged alternately. They are mounted in each corner, near enough to one another that they are bound by covalent (chemical) bonds. A covalent bond is a chemical bond that involves the sharing of electron pairs between atoms.
 Krypton (Kr) is one of the 6 noble gases which appear on the far right of the Periodic Table. The other five are Helium (He), Neon (Ne), Argon (Ar), Xenon (Xe) and Radon (Rn).
Krypton (Kr) is one of the 6 noble gases which appear on the far right of the Periodic Table. The other five are Helium (He), Neon (Ne), Argon (Ar), Xenon (Xe) and Radon (Rn).
Krypton monoxide unit cell is cuboid
Krypton monoxide’s unit cell is cuboid with a diamond base, with krypton atoms at the corners. Additionally, in the middle of the two opposite side walls, there is one krypton atom.
Lata said:
“Where is the oxygen? On the side walls of the unit cell, where there are five atoms of krypton, they are arranged like the dots on a dice showing the number five. Single atoms of oxygen are located between the krypton atoms, but only along the diagonal – and only along one!”
“Thus, on each wall with five krypton atoms there are only two atoms of oxygen. Not only that, the oxygen is not exactly on the diagonal: one of the atoms is slightly offset from it in one direction and the other atom in the other direction.”
In such an idiosyncratic unit cell, each oxygen atom is chemically bound to the two closest adjacent krypton atoms.
Zig-zag chains of Kr/O\Kr\O/Kr will consequently pass through the krypton monoxide crystal, forming long polymer structures. A polymer is a substance which has a molecular structure built up mainly or completely from a large number of similar units bonded together.
According to calculations, crystals of this type of krypton monoxide should have semiconductor characteristics. One would assume that they will be dark and not very transparent.
Less stable krypton compounds also found
Theorists from the Institute of Physical Chemistry have also found a second, slightly less stable krypton compound – the tetroxide KrO4. This material, which probably has metal-like properties, has a simpler crystalline structure and requires a pressure higher than 3.4 million atmospheres for its formation.
After formation, the two types of krypton oxide crystals could likely exist at a lower pressure than that required to make them. One atmosphere – the pressure on Earth at sea level – however, is so low that on our planet’s surface these crystals would undergo immediate degradation.
Dr. Zaleski-Ejgierd said:
“Reactions occurring at extremely high pressure are almost unknown, very, very exotic chemistry. We call it ‘Chemistry on the Edge’. Often the pressures needed to perform syntheses are so gigantic that at present there is no point in trying to produce them in laboratories.”
“In those cases even methods of theoretic description fail! But what is most interesting here is the non-intuitiveness. From the very first to the last step of synthesis you never know what’s going to happen.”
And so the scientists return to their computers where simulations of subsequent syntheses are reaching completion.
Citation: “Krypton oxides under pressure,” Patryk Zaleski-Ejgierd & Pawel M. Lata. Scientific Reports 6, Article number: 18938 (2016). 2nd February, 2016. DOI: 10.1038/srep18938.