Some of the basic components required for life on our planet most likely came from comets and other celestial objects, specifically ribose – a sugar needed to make DNA and RNA – says a team of scientists from France, Mexico and Denmark.
Cornelia Meinert, head of research at the Université Nice Sophia Antipolis in France, and colleagues explained in the journal Science (citation below) that they detected ribose in laboratory experiments that simulated the very early Solar System.
They shone ultraviolet light on a simple, frozen mixture of chemicals that mimicked the ices that form in space between stars – in places like comets and asteroids.
 For life to exist there has to be genetic material (DNA and RNA). For DNA and RNA to exist certain sugars such as ribose must be present. The ingredients to create these sugars most likely came from space, specifically comets and other celestial objects. (Image: twitter.com/connycnrs)
For life to exist there has to be genetic material (DNA and RNA). For DNA and RNA to exist certain sugars such as ribose must be present. The ingredients to create these sugars most likely came from space, specifically comets and other celestial objects. (Image: twitter.com/connycnrs)
As the mixture of chemicals condensed and then warmed up, the ice produced considerable quantities of ribose (C5H10O5), as well as some other interesting molecules including arabinose, xylose, and lyxose.
Sugars can be made in a simple way
The authors say that theirs is the first study to show that sugars can be produced in such a simple manner.
They believe that these critical molecules are likely formed when similar ices condense around dust grains and comets close to a young star.
How complex sugars such as ribose could emerge from the seemingly hostile, icy environment of a solar nebula has been a mystery for scientists. Solar nebula refers to the disc-shaped cloud that existed before our Solar System was formed.
Amino acids and some other building blocks of life have been detected in previous laboratory experiments and also detected in comet and meteorite samples. Sugars, however, have been much more of an enigma, mainly because they are so difficult to detect.
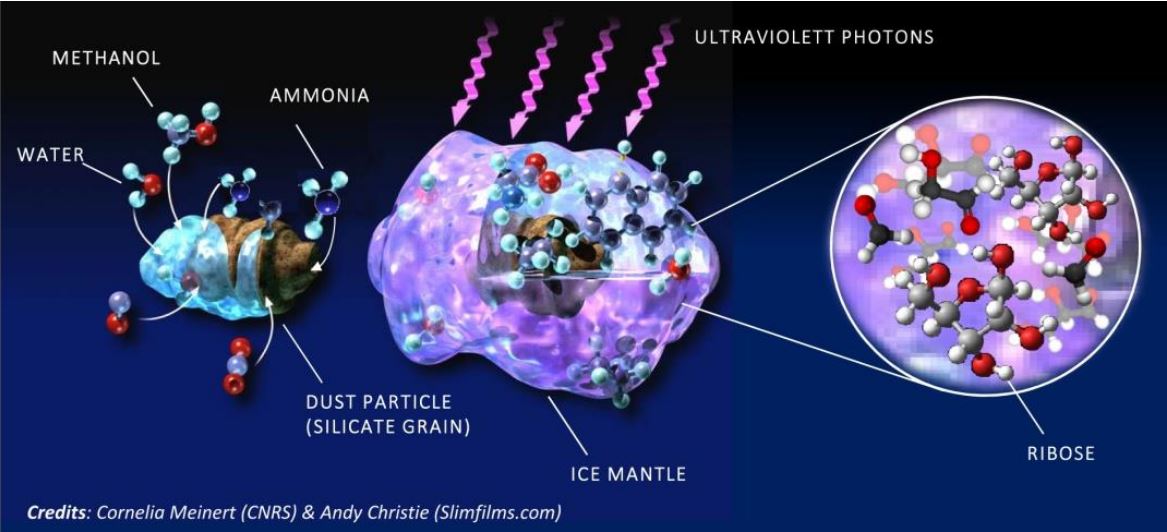 Ribose is formed in the coating of ice around grains of dust, from simple precursor molecules (water, methanol and ammonia) and under the effect of intense radiation. (Image: cnrs.fr/sites/communique)
Ribose is formed in the coating of ice around grains of dust, from simple precursor molecules (water, methanol and ammonia) and under the effect of intense radiation. (Image: cnrs.fr/sites/communique)
Sugars hard to detect
The authors said they would not be surprised if other experiments around the world had also manufactured ribose and other sugars – they may have been there all along without being detected.
The scientists used a sophisticated technique called multidimensional gas chromatography, which was the reason they were able to detect the sugars.
In an interview with BBC News, Dr. Meinert said:
“In all the experiments that were run for the last 20-30 years around the world using preparative HPLC column or other equipment, the sugars were probably there. We have a fancy technique called multidimensional gas chromatography – and this was the reason why we are now able to detect them.”
Dr. Meinert’s team, along with colleagues from the Synchrotron SOLEIL & Institut d’Astrophysique Spatiale, both in France, the Universidad Autónoma del Estado de Morelos in Mexico, and Aarhus University in Denmark, mixed ammonia and methanol with water, and subjected the mixture to low pressure and extremely low temperature (-195°C) in a vacuum chamber.
It was then allowed to condense on a very cold surface, in the same way ‘pre-cometary’ ice might settle around a grain of dust.
They blasted the mixture with intense UV light as it condensed – as would have occurred when exposed to a young Sun – and allowed it to warm up to room temperature.
 Ultraviolet treatment of ‘pre-cometary’ ice (left) reproduced the natural evolution of frozen molecules observed in interstellar clouds (right, the Pillars of Creation), leading to the formation of sugar molecules. (Image: cnrs.fr/sites/communique)
Ultraviolet treatment of ‘pre-cometary’ ice (left) reproduced the natural evolution of frozen molecules observed in interstellar clouds (right, the Pillars of Creation), leading to the formation of sugar molecules. (Image: cnrs.fr/sites/communique)
The resulting residue contained ribose, arabinose, xylose, lyxose and some other complex molecules.
In an Abstract in the journal, the authors wrote:
“Our results suggest that the generation of numerous sugar molecules, including the aldopentose ribose, may be possible from photochemical and thermal treatment of cosmic ices in the late stages of the solar nebula.”
“Our detection of ribose provides plausible insights into the chemical processes that could lead to formation of biologically relevant molecules in suitable planetary environments.”
Lots of organics formed in these ices
Dr. Meinert said:
“You might think that there are not a lot of organics formed in these ices – but in fact it’s the opposite. We see a lot of different compounds and classes of compounds: amino acids, acids, alcohols, aldehydes – and the sugars. This means that the sample is very complex.”
All these compounds could be dissolved in water, which is a necessary state when incorporating them into fledgling life forms.
This study adds further proof to the notion that even though life on Earth most likely started here, its basic ingredients came from outer space. Initially we all, including our planet, the Sun, the Moon, plus all life on Earth, come from stardust.
Andrew Mattioda, an astrochemist at NASA Ames Research Center at Moffett Federal Airfield in California’s Silicon Valley, who was not involved in this latest study, said:
“If all these molecules that are necessary for life are everywhere out in space, the case gets a lot better that you’ll find life outside of Earth.”
Lead author, Uwe Meierhenrich, said the findings also suggest that the genetic material of any life forms on other planets could potentially be based around different sugars:
“We did not only detect ribose, but many other sugar molecules,’ he says. ‘[This] does not necessarily hint to RNA that preceded DNA and evolved from ribose. Alternative nucleotide precursors might have played their role in chemical evolution as well.”
The researchers would like to carry out further studies, and explore other questions such as the chirality of ribose.
Dr. Meierhenrich said:
“We would like to understand the reason for which d-ribose is used in DNA instead of l-ribose. We assume that the reason for this symmetry-breaking event can be found in interstellar space.”
Citation: “Ribose and related sugars from ultraviolet irradiation of interstellar ice analogs,” Cornelia Meinert, Louis Le Sergeant d’Hendecourt, Uwe J. Meierhenrich, Iuliia Myrgorodska, Pierre de Marcellus, Thomas Buhse, Laurent Nahon & Søren V. Hoffmann. Science. 8 April 2016. DOI: 10.1126/science.aad8137.

